top of page
Search


Skin Tone Diversity in Pulse Oximetry Studies: Why Inclusive Participant Recruitment Matters
Why Diversity Matters in Pulse Oximetry Studies Pulse oximeters estimate oxygen saturation using light absorption through the skin. Because optical properties vary among individuals, skin tone diversity in clinical research studies is essential for accurate device evaluation. A well-designed pulse oximetry study must include participants representing a range of skin tones. This ensures that device performance is assessed across physiologically diverse populations and hel
Apr 92 min read

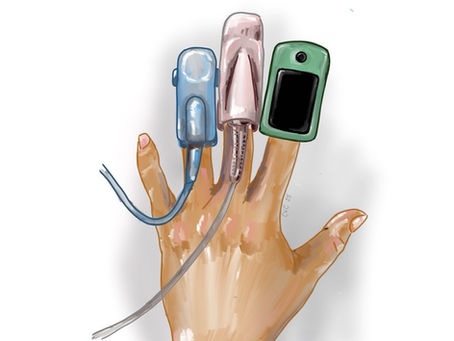
Understanding Transfer Standards in Pulse Oximetry Studies
Understanding Transfer Standards in Pulse Oximetry Studies Pulse oximeter development often begins long before a formal accuracy study is performed. During this stage, engineering teams are refining sensors, algorithms, calibration strategies, and signal-quality controls. To move development forward efficiently, they need a reliable way to compare device performance under controlled conditions before initiating a full pulse oximetry study with arterial blood sampling. This
Apr 23 min read


Designing a Pulse Oximetry Study for Regulatory Success
Well-designed pulse oximetry studies generate regulator-ready datasets for device validation. Regulatory Expectations for Pulse Oximetry Validation Regulators expect robust clinical evidence when evaluating pulse oximeter performance. This evidence must demonstrate device accuracy across a defined range of oxygen saturation levels. To generate this evidence, organizations conduct pulse oximetry validation studies in controlled clinical environments. The physiological data
Mar 312 min read


From Engineering to Clinical Evidence: Supporting Pulse Oximeter Development
Clinical studies help translate pulse oximeter development into validated physiological performance data. Challenges in Pulse Oximeter Development Modern pulse oximeters are complex sensing systems that rely on optical measurements and signal processing algorithms. While laboratory testing can evaluate hardware performance, it cannot fully replicate human physiology. To understand how a device performs during real physiological changes, developers rely on pulse oximetry st
Mar 262 min read


Hypoxia Labs and Their Role in Pulse Oximetry Testing
Specialized hypoxia labs enable controlled oxygen conditions required for pulse oximetry testing. Why Pulse Oximetry Testing Requires Controlled Environments Pulse oximeters estimate oxygen saturation using optical sensors. Validating these measurements requires clinical studies that compare device readings with direct arterial measurements. Because oxygen saturation cannot be reliably adjusted in typical clinical settings, pulse oximetry testing must take place in specia
Mar 242 min read


The Science Behind a Controlled Desaturation Study for Pulse Oximetry Validation
Controlled desaturation protocols allow researchers to evaluate pulse oximeter accuracy across multiple oxygen saturation levels. Why Controlled Desaturation Is Essential for Pulse Oximetry Studies A pulse oximetry study evaluates how accurately a device estimates arterial oxygen saturation. To measure this performance reliably, researchers must observe device readings across a range of oxygen levels. This is achieved through a controlled desaturation study , where oxygen
Mar 193 min read


Pulse Oximetry Studies: Methods, Standards, and Clinical Validation in Hypoxia Labs
Why Pulse Oximetry Studies Matter for Device Validation Pulse oximeters play a critical role in physiological monitoring, but demonstrating their accuracy requires carefully designed clinical research studies. Optical sensing systems must be validated against direct measurements of arterial oxygen saturation to confirm their performance across a wide range of oxygen saturations. A well-designed pulse oximetry study allows researchers to compare device reported oxygen satu
Mar 174 min read


Understanding Transfer Standards in Pulse Oximetry Development
Pulse oximeter development often requires dependable performance feedback well before a formal regulatory accuracy study is initiated. During this stage, teams iterate on sensor hardware, algorithms, calibration curves, and signal-quality controls. To make meaningful progress, they need a repeatable way to compare performance across prototypes and study sessions. A transfer standard helps fill that role. It is an intermediary pulse oximeter system with calibration traceable
Mar 113 min read


FDA General Wellness Policy: What Non-Invasive Sensing Products Must Meet to Remain Low Risk
As wearable and software-enabled technologies increasingly estimate physiological parameters such as oxygen saturation, blood pressure, and heart rate variability, regulatory classification has become a critical design consideration. The FDA’s General Wellness: Policy for Low Risk Devices clarifies when products using non-invasive sensing—such as optical sensing—may be considered general wellness products rather than regulated medical devices. For developers conducting a cl
Feb 163 min read
bottom of page

