top of page
Search
All Posts


Korotkoff Sounds and Dual Auscultation: The Reference Behind Blood Pressure Validation
Blood pressure validation studies depend on a reference method that is clinically grounded and repeatable. For intermittent blood pressure devices, that reference is often manual auscultation, where trained observers use a cuff and stethoscope to determine systolic and diastolic pressure based on Korotkoff sounds. These sounds provide the clinical signal used to identify the points at which blood flow first returns and then becomes silent again as the cuff deflates. For inte
May 193 min read


IEEE 1708-2025 Overview: Study Design for Cuffless Blood Pressure Technologies
Why IEEE 1708-2025 Matters for Cuffless Blood Pressure Validation Cuffless blood pressure technology is advancing quickly, and credible performance claims depend on well-designed clinical validation studies. Unlike traditional cuff-based monitors, wearable and cuffless devices may use sensors, algorithms, calibration methods, or continuous signal processing to estimate blood pressure. IEEE 1708-2025 establishes an evaluation framework for wearable cuffless blood pressure me
May 143 min read


ISO 81060-3 Clinical Validation for Continuous Blood Pressure Monitoring
Why ISO 81060-3 Matters for Continuous Blood Pressure Devices Continuous non-invasive blood pressure monitoring is fundamentally different from intermittent cuff readings. Instead of producing a single systolic and diastolic value at discrete time points, continuous systems output frequent blood pressure estimates and trends over time. ISO 81060-3 defines the clinical validation framework for evaluating continuous non-invasive blood pressure devices under standardized condi
May 122 min read


Designing a Blood Pressure Validation Study: Protocols and Reference Methods
Planning a Blood Pressure Validation Study Designing a blood pressure validation study requires careful planning to support reliable measurement comparisons. Early clinical trial protocol development should clearly define the device’s intended use, primary accuracy endpoints, and reference method used for comparison. This up-front structure helps keep study execution, data collection, and reporting consistent from the first participant through the final report. Key Eleme
May 72 min read


ISO 81060-7 and FDA Guidance for Wearable Blood Pressure Devices
The Rise of Wearable Blood Pressure Devices Advances in sensor technology and signal processing have accelerated the development of wearable and cuffless blood pressure devices, including systems designed for intermittent spot checks and those that aim to estimate blood pressure more frequently over time. Unlike traditional cuff-based monitors, wearable and cuffless technologies must perform under real-world conditions where movement and posture can influence measurements.
May 52 min read


ISO 81060-2 Blood Pressure Device Validation Explained
What Is ISO 81060-2? ISO 81060-2 is an international standard that defines how to clinically validate intermittent automated non-invasive blood pressure devices. It establishes the study framework used to determine whether a device’s systolic and diastolic readings align with qualified reference measurements under standardized conditions. Key Components of the Standard Validation studies conducted under ISO 81060-2 typically include: Structured measurement sequences using
Apr 301 min read


Arterial Line Reference Measurements in Blood Pressure Studies
The Importance of High-Resolution Reference Measurements Blood pressure validation studies require reference measurements that accurately reflect physiological conditions. While auscultatory measurements provide reliable values, some studies require continuous high-resolution monitoring. In these cases, researchers may use arterial line monitoring. What Is Arterial Line Monitoring? An arterial catheter allows researchers to measure blood pressure directly within an art
Apr 281 min read


Dual Auscultation in Blood Pressure Validation Studies
Why Reference Measurements Matter Every blood pressure validation study relies on a reliable reference method to evaluate device accuracy. A trusted reference measurement provides the foundation for determining whether a monitoring device delivers accurate and clinically meaningful readings. At Parameters Research Laboratory (PRL), reference measurements are conducted by highly trained clinical research staff with deep experience in physiological monitoring studies. This lev
Apr 232 min read


Blood Pressure Device Validation Studies: Methods, Standards, and Clinical Research Design
Blood Pressure Device Validation Studies Accurate blood pressure measurement is fundamental to physiological monitoring. As new technologies expand beyond traditional cuffs into wearable and cuffless blood pressure devices, sponsors must demonstrate performance using a structured blood pressure validation study. These studies evaluate how closely a device’s measurements align with reference measurements obtained through established clinical methods and generate the evidence r
Apr 215 min read


Human Factors in Physiological Monitoring Device Validation
Why Human Factors Matter Accuracy alone does not determine whether a physiological monitoring device performs well in real-world use. Human factors studies evaluate how users interact with a device during normal operation. They confirm that the device can be used correctly and repeatedly under the conditions reflected in its claims. For medical device manufacturers, this work strengthens the evidence package by showing that performance is achievable under realistic workflo
Apr 152 min read


Multi-Site Clinical Research Studies for Physiological Monitoring Devices
Why Multi-Site Studies Matter Physiological monitoring devices increasingly rely on clinical evidence that reflects real-world use and diverse populations. Some studies may require clinical data to be collected across multiple research locations. Multi-site clinical research studies allow sponsors to expand recruitment and collect larger datasets while maintaining consistent study protocols. When executed under Good Clinical Practice (GCP) and ISO 14155, multi-site program
Apr 142 min read


Avoiding FDA Submission Delays in Medical Device Validation
Why Submission Delays Occur Regulatory submissions for physiological monitoring devices often depend on clinical evidence demonstrating device performance. When study protocols are poorly aligned with regulatory expectations, organizations may encounter FDA submission delays . Common causes include: incomplete clinical datasets inconsistent study protocols inadequate documentation The Role of Clinical Trial Protocol Development A well-structured clinical trial pro
Apr 101 min read
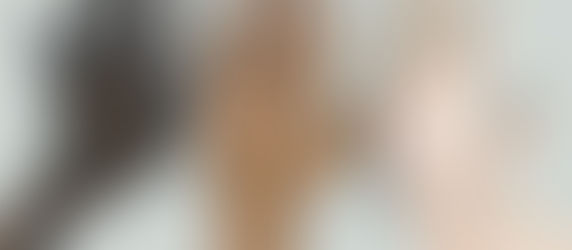
Skin Tone Diversity in Pulse Oximetry Studies: Why Inclusive Participant Recruitment Matters
Why Diversity Matters in Pulse Oximetry Studies Pulse oximeters estimate oxygen saturation using light absorption through the skin. Because optical properties vary among individuals, skin tone diversity in clinical research studies is essential for accurate device evaluation. A well-designed pulse oximetry study must include participants representing a range of skin tones. This ensures that device performance is assessed across physiologically diverse populations and hel
Apr 92 min read


Why Arterial Sampling Matters in Pulse Oximetry Validation
The Importance of Reference Measurements Every pulse oximetry study relies on a reliable reference measurement to evaluate device accuracy. While pulse oximeters estimate oxygen saturation noninvasively, the true arterial oxygen saturation must be determined using blood gas analysis. For this reason, arterial sampling plays a critical role in pulse oximetry validation . How Arterial Sampling Works in a Hypoxia Lab During a controlled desaturation study , participant
Apr 71 min read


Understanding Transfer Standards in Pulse Oximetry Studies
Understanding Transfer Standards in Pulse Oximetry Studies Pulse oximeter development often begins long before a formal accuracy study is performed. During this stage, engineering teams are refining sensors, algorithms, calibration strategies, and signal-quality controls. To move development forward efficiently, they need a reliable way to compare device performance under controlled conditions before initiating a full pulse oximetry study with arterial blood sampling. This
Apr 23 min read


Designing a Pulse Oximetry Study for Regulatory Success
Well-designed pulse oximetry studies generate regulator-ready datasets for device validation. Regulatory Expectations for Pulse Oximetry Validation Regulators expect robust clinical evidence when evaluating pulse oximeter performance. This evidence must demonstrate device accuracy across a defined range of oxygen saturation levels. To generate this evidence, organizations conduct pulse oximetry validation studies in controlled clinical environments. The physiological data
Mar 312 min read


From Engineering to Clinical Evidence: Supporting Pulse Oximeter Development
Clinical studies help translate pulse oximeter development into validated physiological performance data. Challenges in Pulse Oximeter Development Modern pulse oximeters are complex sensing systems that rely on optical measurements and signal processing algorithms. While laboratory testing can evaluate hardware performance, it cannot fully replicate human physiology. To understand how a device performs during real physiological changes, developers rely on pulse oximetry st
Mar 262 min read


Hypoxia Labs and Their Role in Pulse Oximetry Testing
Specialized hypoxia labs enable controlled oxygen conditions required for pulse oximetry testing. Why Pulse Oximetry Testing Requires Controlled Environments Pulse oximeters estimate oxygen saturation using optical sensors. Validating these measurements requires clinical studies that compare device readings with direct arterial measurements. Because oxygen saturation cannot be reliably adjusted in typical clinical settings, pulse oximetry testing must take place in specia
Mar 242 min read


The Science Behind a Controlled Desaturation Study for Pulse Oximetry Validation
Controlled desaturation protocols allow researchers to evaluate pulse oximeter accuracy across multiple oxygen saturation levels. Why Controlled Desaturation Is Essential for Pulse Oximetry Studies A pulse oximetry study evaluates how accurately a device estimates arterial oxygen saturation. To measure this performance reliably, researchers must observe device readings across a range of oxygen levels. This is achieved through a controlled desaturation study , where oxygen
Mar 193 min read

Pulse Oximetry Studies: Methods, Standards, and Clinical Validation in Hypoxia Labs
Why Pulse Oximetry Studies Matter for Device Validation Pulse oximeters play a critical role in physiological monitoring, but demonstrating their accuracy requires carefully designed clinical research studies. Optical sensing systems must be validated against direct measurements of arterial oxygen saturation to confirm their performance across a wide range of oxygen saturations. A well-designed pulse oximetry study allows researchers to compare device reported oxygen satu
Mar 174 min read
bottom of page

